 Molecular nitrogen,? N 2 is a stable (that is relatively unreactive) molecular compound.Ī common N-containing molecule is ammonia (NH 3), which is analogous to methane (CH 4). The two N atoms are bonded together by a triple bond, consisting of a σ and two π bonds. The simplest compound of nitrogen is molecular nitrogen, N 2. As we will see later on elements in the next row, such as phosphorus (P) and sulfur (S), are larger and have more available orbitals for bonding – they can form up to six centers of electron density. Together these factors mean that nitrogen, and in fact all elements in the second row of the periodic table (periodic table tutorial), are limited to bonding arrangements with no more than four centers of electron density. Another factor is that there are only four orbitals available in nitrogen in the second quantum shell - if nitrogen were to form five bonds it would have to use orbitals from the next quantum shell (3), but these orbitals are too high in energy - the energy required would not be offset the energy released upon on bond formation. One factor is that nitrogen atoms are too small to support five centers of electron density around themselves - the bonds begin to overlap, which is destabilizing, just like we saw with bulky groups around a carbon. We can explain this observation in several ways. So if you are following the rules, you might well assume that nitrogen would be able to form five bonds (after all, it has five valence electrons.) But when we look carefully, we NEVER see a nitrogen atom making five bonds - most of the time it makes three bonds! 
Nitrogen has seven electrons (2 core and 5 valence)(1s2, 2s2, 2px1, 2py1, 2pz1). 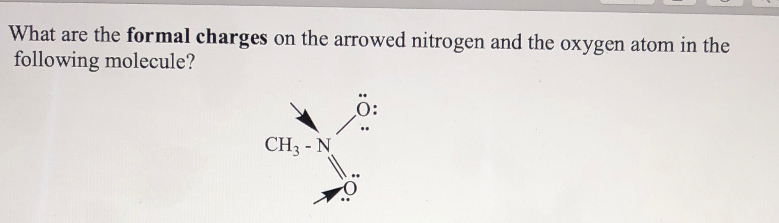
Carbon ?has six electrons (2 core and 4 valence), and can form four bonds with neighboring atoms. From the perspective of living systems, two of the most interesting elements are nitrogen and oxygen. There are many other elements, and their properties add chemical complexity to molecular behavior. While the bonding of H and C atoms can generate a remarkable array of molecules, the hydrocarbons are really rather boring (chemically), that is, they take part in a rather limited range of reactions and would not, on their own, be expected to produce anything like life. 
Chemistry, life, the universe and everything Chapter 4.4: Bonding in Nitrogen, Oxygen & Fluorine 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
